Good Clinical Practice Inspections
Understand the processes for preparation, conduct and follow up of Good Clinical Practice Inspections.
Implementation of ICH E6 (R3) Guideline for Good Clinical Practice (GCP)
The ICH guideline for GCP is an international ethical and scientific quality standard for designing, conducting, recording and reporting clinical trials. Compliance with GCP provides assurance that the rights, safety and well-being of trial participants are protected, and clinical trial data are credible, facilitating mutual acceptance of the clinical data by regulatory authorities. The E6 (R3) guideline addresses GCP principles in the context of an evolving landscape of clinical trials, supporting innovations in clinical trial design, operational approaches, and technological advancements. It promotes a quality-by-design and risk-proportionate approach to clinical trial conduct.
The Health Sciences Authority (HSA) has adopted the Principles and Annex 1 of the ICH E6 (R3) Guideline for GCP, which has been implemented in Singapore on 1 Jan 2026.
To ensure a smooth transition to the ICH E6 (R3) Guideline for GCP, interested parties should:
thoroughly review the ICH E6 (R3) Guideline for GCP;
perform a gap analysis and implement necessary changes to align with the new guideline; and
adopt a proportionate approach to training on the ICH E6 (R3) Guideline for GCP and document the relevant training completed.
The Principal Investigator (PI) should be familiar with the ICH E6 (R3) Guideline for GCP, since the PI is overall responsible for trial conduct at the investigator site. The PI should ensure that persons or parties assisting in the clinical trial receive training appropriate to their delegated trial activities that go beyond their usual training and experience, and are adequately informed about the relevant aspects of the ICH E6 (R3) Guideline for GCP.
The proportionate approach to training on the ICH E6 (R3) Guideline for GCP also applies to sponsors.
Refer to the following for more information:
Presentations - ICH E6 (R3) Guideline for GCP
Overview
A Good Clinical Practice (GCP) Inspection is an official review of documents, facilities, records and any other resources that are related to your clinical trial. The inspection may be done at the site of the trial, the sponsor's or contract research organisation's facilities or at another appropriate location. The inspection may also take place at multiple sites where required.
The following types of clinical trials may be subjected to GCP Inspections:
Clinical trials of therapeutic products and Class 2 cell, tissue or gene therapy products (CTGTPs) that are subject to the requirements of a Clinical Trial Authorisation (CTA) or Clinical Trial Notification (CTN)
Clinical trials of medicinal products that are subject to the requirements of a Clinical Trial Certificate (CTC)
The inspectee of a GCP Inspection may either be the Principal Investigator or the sponsor of the clinical trial being inspected.
Objectives of GCP Inspections
GCP inspections may either be protocol-specific or systems. The objectives of both types of inspections are to:
Safeguard the rights, safety and well-being of trial participants
Verify the quality and integrity of clinical trial data submitted to the regulatory authorities
Assess compliance to protocol, applicable regulations, guidelines and standard operating procedures
Additionally, the objectives of systems inspections are to:
Assess whether a system is suitably designed, controlled, maintained and documented to fulfil intended objectives
Identify areas for quality improvement
Types of GCP Inspections
Types of GCP Inspections
Type | Description |
|---|---|
Routine | Usually apply to ongoing clinical trials and are done announced. |
Triggered | Conducted as a result of requests, complaints or reports to us on suspected violations of regulations. Such types of inspections may be done announced and apply to ongoing or completed clinical trials. |
Pre-marketing approval application | Usually apply to completed clinical trials and are done announced. |
GCP Inspection process
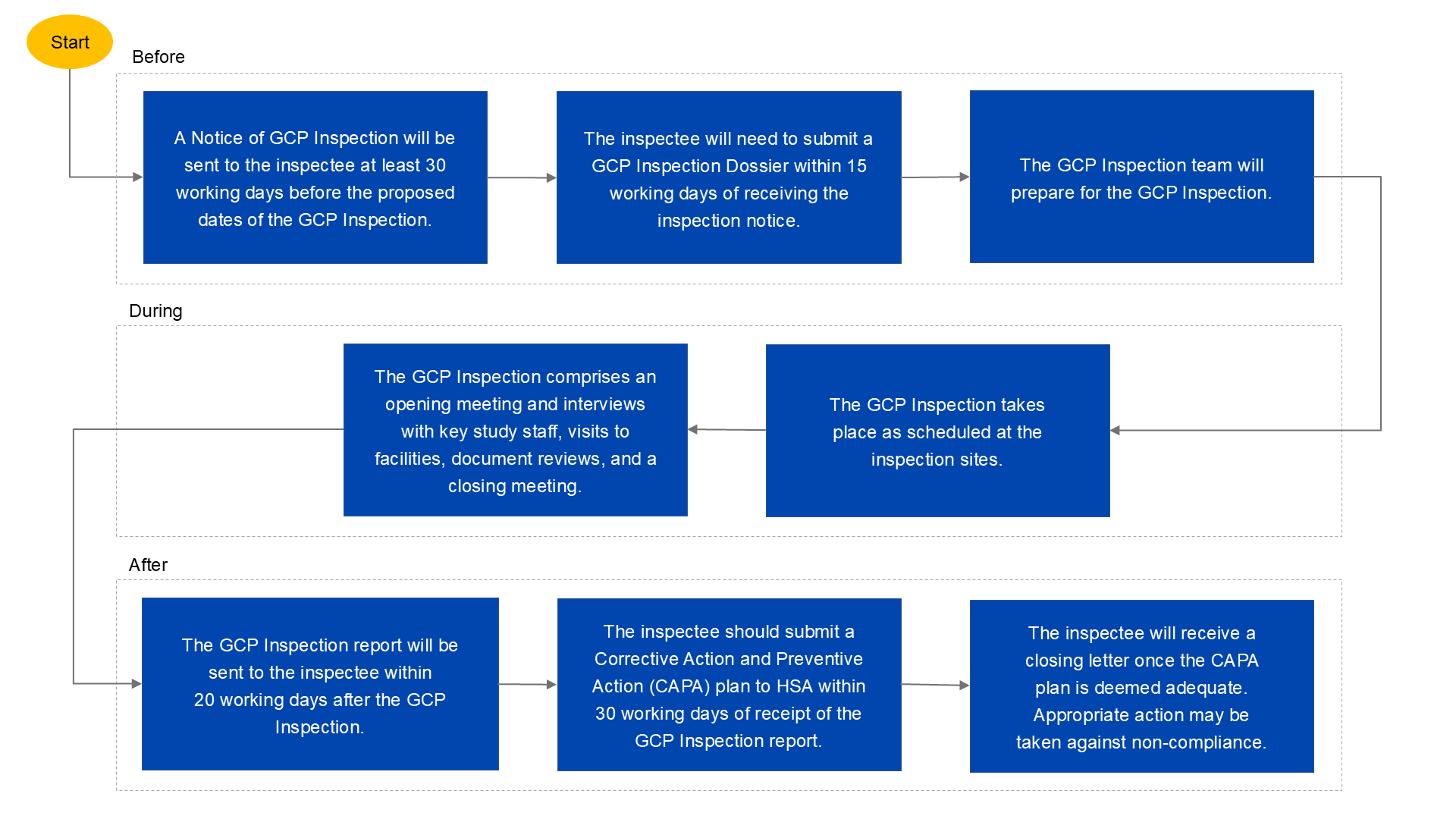
Details of a GCP Inspection
During a GCP Inspection | Details |
|---|---|
Opening meeting | GCP inspectors will explain the GCP Inspection framework, confirm the agenda and confirm that all resources, documents and facilities required for inspection are available. |
Interviews with study staff | Interviews will be conducted with key study staff to determine how the clinical trial is being conducted. |
Visit to facilities | GCP inspectors may visit the site or facilities where the trial is conducted. |
Document review | Essential documents will be reviewed. |
Interim meeting | Observations from the GCP Inspection may be shared to provide the inspectee with an opportunity to clarify or resolve the observations during the GCP Inspection, where possible. |
Closing meeting | Observations from the GCP Inspection will be shared. |
Grading of GCP Inspection Findings
GCP Inspection Findings will be graded based on the impact on the goals of GCP, frequency of occurrence, and corrective and preventive actions that can be taken.
Grading of GCP Inspection Findings
Grading | Impact on the goals of GCP |
|---|---|
Critical | Adversely affects |
Major | Might adversely affect |
Other | Not expected to adversely affect |
Comments | The observations might lead to suggestions on how to improve quality or reduce the potential for a non-compliance to occur in the future. |
Remote / Hybrid GCP Inspections
Based on the experience gained from the COVID-19 pandemic, remote / hybrid approaches to GCP inspections may be incorporated into regular GCP inspections where appropriate and feasible, based on the inspection scope and if the objectives of GCP Inspection will not be compromised.
Refer to the Guidance on GCP Compliance Inspection Framework [PDF, 415 KB] for more details on the GCP Inspection.
Presentations
Presentations - ICH E6 (R3) Guideline for GCP
DATE | PRESENTATION |
|---|---|
28 Feb 2025 | ICH E6 (R3) Guideline for GCP: Principles and Annex 1 - Overview of Major Changes [PDF, 2.1 MB] Presented in HSA-SCRI Public Webinar |
22 Jan 2025 | ICH E6 (R3) Guideline for GCP: Annex 2 - Overview of Step 2 Draft [PDF, 550 KB] Presented in SCRP Forum on Navigating the Future of Clinical Trials |
Presentations - Common GCP Inspection Findings
DATE | PRESENTATION |
|---|---|
6 Feb 2026 | Looking Back at 2025 [PDF, 1.7 MB] Presented in NHG CRCS Forum |
21 Feb 2025 | Looking Back at 2024 [PDF, 519 KB] Presented in SCRI-NHG CRC Appreciation Day 2025 |
26 Jan 2024 | Looking Back at 2023 [PDF, 991 KB] Presented in CRCS Forum |
6 Jan 2023 | Looking Back at 2022 [PDF, 594 KB] Presented in CRCS Forum |
20 Jan 2022 | Looking Back at 2020-2021 [PDF, 1.3 MB] Presented in CRCS Forum |
3 Dec 2019 | Looking Back at 2019 [PDF, 475 KB] Presented in CRCS Forum |
7 Dec 2018 | Common GCP Inspection Findings in 2018 [PDF, 428 KB] Presented in CRCS Forum |
1 Dec 2017 | Common GCP Inspection Findings in 2017 [PDF, 595 KB] Presented in Combined CRP-CRCS Forum |
16 Jan 2017 | |
3 Dec 2015 | Common GCP Inspection Findings in 2015 [PDF, 498 KB] Presented in Combined CRP-CRCS Forum |
12 Dec 2014 | Common GCP Inspection Findings in 2014 [PDF, 1.1 MB] Presented in Combined CRP-CRCS Forum |
13 Nov 2013 | Common GCP Inspection Findings in 2013 [PDF, 902 KB] Presented in Combined CRP-CRCS Forum |
2012 | |
2011 | |
2010 |
