Regulatory overview of cell, tissue and gene therapy products
Understand the scope of CTGTP regulations in Singapore.
What are cell, tissue and gene therapy products (CTGTP)?
CTGTP are health products intended for use in humans for a therapeutic, preventive, palliative or diagnostic purpose. CTGTP can contain any of the following and achieves its primary intended action by pharmacological, immunological, physiological, metabolic or physical means:
viable or non-viable human cells or tissues
viable animal cells or tissues
recombinant nucleic acids
Is my product a Cell, Tissue and Gene Therapy Product?
Answer a few questions in our self-help tool to find out if your product is considered a CTGTP in Singapore.
The following products are NOT considered CTGTP:
Recombinant vaccines for a preventive purpose. Such products are typically considered therapeutic products instead.
In-vitro diagnostic products. Such products are typically considered medical devices instead.
Cells and tissues obtained from a patient that are minimally manipulated and reimplanted into the same patient for homologous use during the same surgical procedure (e.g., coronary artery bypass graft)
Bone marrow, peripheral blood or umbilical or placental cord blood from a human that is minimally manipulated and intended for homologous use
Organs and tissues that are minimally manipulated and intended for transplant
Reproductive cells (sperm, eggs) and embryos intended for assisted reproduction
Whole blood any blood component that is minimally manipulated and intended for treating blood loss or blood disorders
Products 4 to 7 are regulated by the Ministry of Health under the Healthcare Services Act (HCSA).
Legislation
We regulate CTGTP in Singapore under the Health Products Act (HPA) and its regulations, including the Health Products (Cell, Tissue and Gene Therapy Products) Regulations 2021.
In Singapore, CTGTP are risk-stratified into two classes below:
CTGTP Classification in Singapore
Class 1 CTGTP (lower risk) | Class 2 CTGTP (higher risk) |
CTGTP which satisfies ALL the following criteria:
| Other CTGTP which are not classified as Class 1 CTGTP. |
Examples of Class 1 CTGTP:
| Examples of Class 2 CTGTP:
|
^Minimally manipulated refers to any processing of the cell or tissue stated below that does not alter the cell's biological characteristics or functions, or the tissue's structural properties: cutting or sizing, grinding, shaping, centrifugation, soaking in an antibiotic or antimicrobial solution, sterilisation or irradiation, cell separation, concentration or purification, filtration, lyophilisation, freezing, cryopreservation or vitrification.
*Homologous use refers to using the CTGTP to repair, reconstruct, replace or supplement the cells or tissues of the recipient to perform the same basic function(s) as the original cells and tissue in the donor in the same anatomical or histological environment. E.g., bone grafts for orthopaedic indications.
Scope of regulation
Manufacturers, importers, registrants and/or suppliers (including wholesalers) of all CTGTP are required to fulfil the relevant duties and obligations of dealings in CTGTP
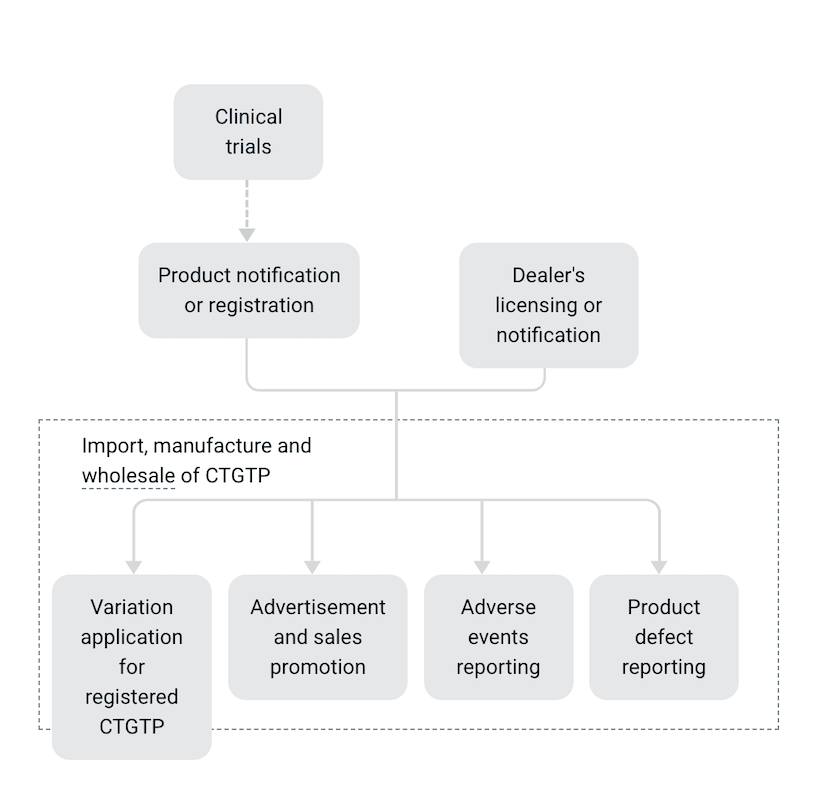
Clinical trials
Clinical trials involving Class 1 CTGTP are regulated under the Human Biomedical Research Act (HBRA) by the Ministry of Health. However, the manufacture, import and supply of CTGTP used as a clinical research material (CRM) in Singapore must comply with the regulations of CRM under the Health Products Act.
For Class 2 CTGTP, you may be required to apply for a Clinical Trial Authorisation or submit a Clinical Trial Notification to us before conducting the clinical trials in Singapore.
Product notification or registration
Before you can supply a CTGTP in Singapore, you will have to first:
Submit a product notification for Class 1 CTGTP to HSA and receive our acceptance of the notification.
Register your Class 2 CTGTP. Registration requirements will differ depending on your chosen registration evaluation route.
Dealer's licensing or notification
Before you can import, manufacture and supply CTGTP in Singapore, you will need to first:
Submit a dealer’s notification if you are dealing in minimally manipulated CTGTP.
Apply for a dealer's licence if you are dealing in more than minimally manipulated CTGTP.
Variation application for registered CTGTP
Submit a variation application to us when there is a change to your registered Class 2 CTGTP safety, efficacy or quality information.
Advertisement and sales promotion
You do not need to obtain prior approval from us before you advertise or promote your CTGTP.
However, you must comply with our regulations regarding advertising claims of CTGTP. Advertising and promotional messages should not contain misleading claims, lead to improper use of the product, or bring undue harm to the public.
Adverse events reporting
If the CTGTP you are marketing poses a serious threat to health, causes death, or undesirable effects, you must report the adverse event to us within a given time frame.
Product defect reporting
As a dealer or product registrant, you are responsible for the safety, quality and efficacy of your CTGTP, and must know how to report and recall defective products that may potentially cause harm to patients or the public.
