What is Digital Health
Digital health includes diverse categories of products comprising telehealth and telemedicine, mobile health, wearable devices, health information technologies and personalised medicine.
It refers to the usage of connected devices, wearables, software including mobile applications (apps) and artificial intelligence (AI) to address various health needs via information and communications technologies.
Digital health has opened up the medical device space to an array of providers such as software or mobile app developers and IT solution providers.
Supporting Digital Health Product Innovation
1. Regulatory Guidelines for Telehealth Products
These guidelines were developed in 2017 to help manufacturers, developers or importers of a digital health device or tool i) to determine if their device, software or app are regulated medical devices under HSA and ii) understand the relevant regulatory requirements.
2. Immediate Registration Pathway for Standalone Software and Mobile Applications
This pathway was implemented in 2018 by leveraging the regulatory review and approval from our reference regulatory agencies in Australia, Canada, the European Union, Japan and the United States. This pathway allows immediate market access upon successful submission of a product registration application, while we perform a backend review to verify the qualification criteria are met and that these devices are safe and effective for use on our patients. More information on the various product registration pathways can be accessed in our Guidance on medical device product registration.
3. Device Development Consultation Scheme
Under this scheme, we provide consultation to researchers, developers and manufacturers of digital health devices, software, apps and AI solutions. To date, digital health products constitute over 40% of these consultations.
4. Regulatory Guidelines for Software Medical Devices
These guidelines were published in April 2020 to mitigate digital threats such as cybersecurity, data integrity, and data security. This serves as a one stop reference on the regulatory requirements for management of software in medical devices throughout its entire life cycle.
5. Artificial Intelligence (AI) in Healthcare Guidelines
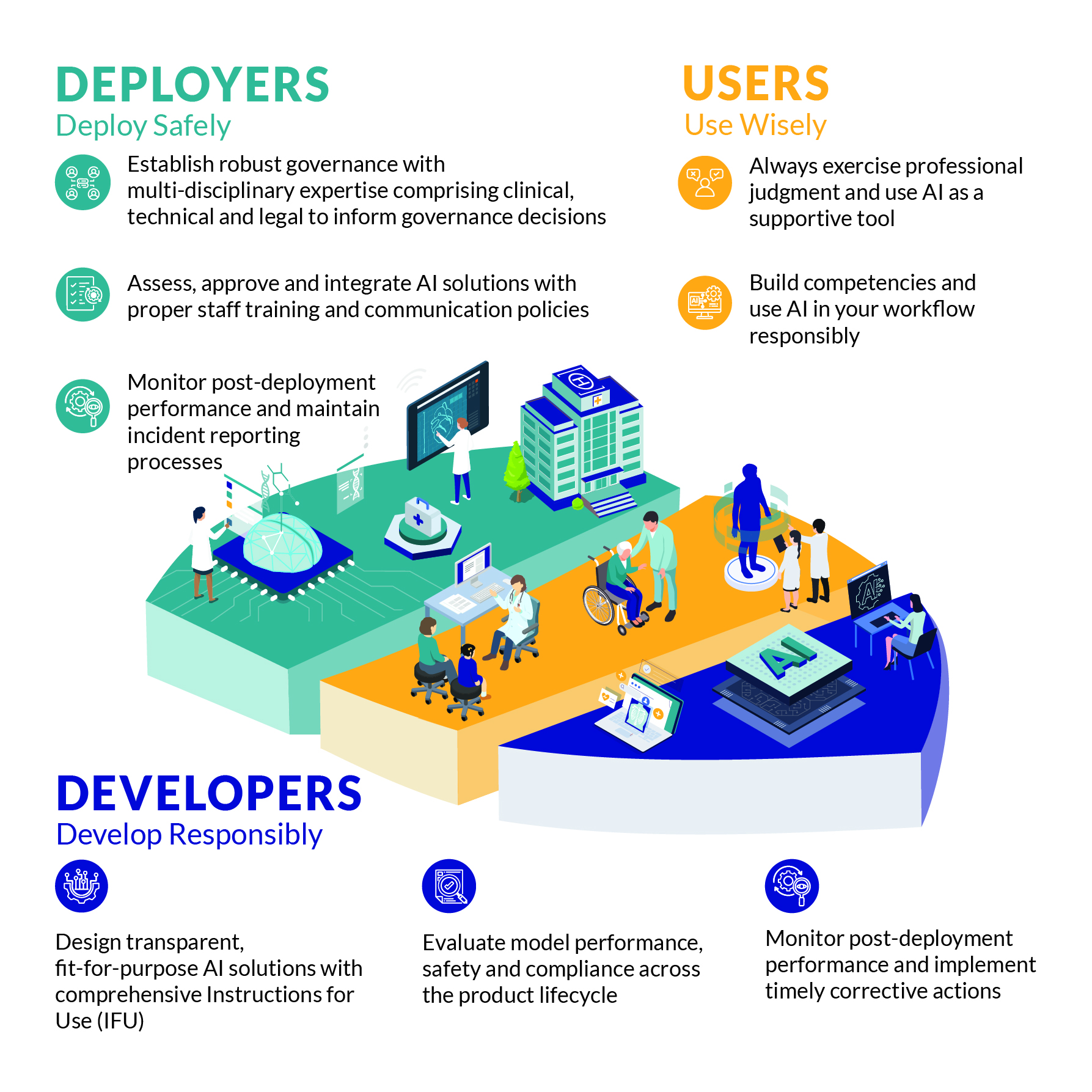
Singapore is committed to adopt AI thoughtfully, prioritising patient safety and clinical effectiveness. MOH continues to develop guidelines to support the safe and responsible use of AI in the healthcare sector. Updated jointly by MOH and the Health Sciences Authority (HSA), the Artificial Intelligence in Healthcare Guidelines (AIHGle 2.0) (read as 'agile') clarifies that AI should augment and empower our healthcare professionals in enhancing healthcare delivery, with patients at the heart of it.
Building on the 2021 framework, AIHGle 2.0 provides practical guidance to support the safe development, deployment and use of AI in healthcare, benefitting patients and improving trust. It also complements HSA's Regulatory Guidelines for Software as Medical Devices. Key updates include:
- Strengthening accountability through clarity of responsibilities for each stakeholder group - developers (e.g. manufacturers), deployers (i.e. healthcare organisations), and users (i.e. healthcare professionals);
- Improving trust via guidance on transparency to facilitate informed decision-making; and
- Updated guidance on AI deployment, such as assessing and mitigating risks.
AIHGle is a living document that will be periodically updated to provide appropriate guidance in light of developments and new technologies. Let us work together to enable safe and trusted AI across the healthcare ecosystem.
6. Supporting Digital Health Product Innovation
The Ministry of Food and Drug Safety (MFDS) Korea and the Health Sciences Authority (HSA) Singapore have collaboratively released guiding principles for conducting clinical trial for machine learning-enabled medical device (MLMD) . The purpose of these guiding principles is to address the unique challenges posed by MLMD in clinical studies. The MFDS and HSA aim to facilitate the development and assessment of MLMD, ensuring that they meet rigorous standards for safety and effectiveness.
Identifying Digital Health Products that are Medical Devices
As a general rule, a digital health device intended for medical purposes such as investigation, detection, diagnosis, monitoring, treatment or management of any medical condition, disease, anatomy or physiological process; will be classified as a medical device subject to HSA’s regulatory controls.
Examples of medical devices which incorporate digital health technology are as follows: