HSA Joins INTERPOL in Global Clampdown on Illegal Health Products
About 1,000 online listings and 6,600 units of illegal health products removed by HSA in joint international operation
As part of an INTERPOL-led global enforcement exercise named Operation Pangea XVIII, the Health Sciences Authority (HSA) took down 959 illegal health product listings from local e-commerce and social media platforms, and issued warnings to 152 sellers. HSA also seized 6,641 units of illegal health products from the borders that were intercepted at Singapore’s land, air and sea checkpoints.
2 Ninety countries participated in the international enforcement exercise which was held over two weeks from 10 March to 23 March 2026. Illicit health products such as unregistered medicines, medical devices and adulterated herbal medicines, as well as pharmaceutical manufacturing equipment were targeted for enforcement actions. HSA has participated in every edition of this collaborative effort since the inception of Operation Pangea in 2008.
3 In Singapore, the listings removed during the latest Operation Pangea included a wide range of products such as unregistered contact lenses, prescription-only skin creams for acne and eczema, sexual enhancement medicines, nasal aspirators and nasal sprays used for relief of nasal congestion and allergies, and oxygen concentrators. Unregistered contact lenses accounted for over 82% of the listings removed. HSA would like to remind sellers that it is illegal to sell contact lenses online. Contact lenses are medical devices that must be registered with HSA to ensure that they meet the required standards of safety, performance and quality. There have been local cases of severe eye injuries from use of contact lens purchased online. Consumers are therefore required to undergo eye examination and contact lens fitting by registered optometrists or contact lens practising opticians.
Intensified enforcement at the borders during two-week Operation
4 In addition to the online listings that were removed, HSA seized 6,641 units of illegal health products at the borders, with the majority (88%) intercepted via postal services. The bulk of the products seized at the borders were prescription medicines such as painkillers or sedatives (about 36%), anti-parasitic medicines such as ivermectin (30%), and dermal fillers (about 6%). These products were stopped in time before they could reach the public and potentially cause harm. Examples of the seized health products included ivermectin and dermal fillers (refer to Annex for photos and infographic).
Ivermectin
5 HSA has stopped members of the public who had tried to import or use ivermectin for self-medication. Ivermectin is a prescription-only medicine registered in Singapore for the treatment of parasitic worm infections. It is not an anti-viral medicine. During the COVID-19 pandemic, there were cases of imports of ivermectin. HSA would like to remind that ivermectin is not approved for use in the prevention or treatment of COVID-19. Self-medicating with ivermectin can be dangerous to your health. There have been local reports of patients requiring hospitalisation after self-medication with ivermectin. Other side-effects include vomiting, diarrhoea, stomach pain, neurologic adverse events (dizziness, seizures, confusion), sudden drop in blood pressure, severe skin rash potentially requiring hospitalisation, and liver injury (hepatitis). Ivermectin can also interact with other medications such as blood-thinners. Consumers are strongly advised not to self-medicate with ivermectin and to consult their doctor for proper treatment of COVID-19.
Dermal fillers
6 110 boxes containing pre-filled syringes of dermal fillers were also seized during Operation Pangea XVIII. These pre-filled syringes could have caused serious harm if they had not been intercepted. Investigations are ongoing.
7 HSA reminds the public that products such as dermal fillers must only be administered by registered healthcare professionals under strict conditions. Those sold online for self-administration may not be manufactured under proper conditions or may contain unknown, unverified and potentially harmful ingredients. There are risks of exposure to toxic chemicals and infectious organisms when injected into the face or body. They can also cause scarring and asymmetric appearance if administered inappropriately. Serious adverse complications associated with the use of such injectable products can include anaphylactic shock (life-threatening allergic reaction), severe infections, blockage of blood vessels leading to tissue death, blindness and stroke. Investigations are ongoing for the case on dermal fillers.
Advisory
8 HSA would like to remind the public to be cautious when products are sold at prices or health claims “too good to be true”. Sellers may adopt promotional tactics to bait consumers into buying illegal products, creating a sense of urgency on not missing out special deals and cheap offers. The use of illegal health products, such as unregistered medical devices and illegal medicines, may not offer the expected product efficacy or could even harm one’s health due to adverse effects arising from adulterated or undeclared contents.
9 Everyone has a part to play in protecting our safety and health through providing feedback on suspected adulterated and harmful products and reporting of adverse events. Public vigilance and reporting help HSA to act swiftly on cases of illegal health products and protect the public against further harm.
10 HSA Chief Executive Officer, Adjunct Professor (Dr) Raymond Chua said: "We urge the general public, healthcare professionals and the industry to continue to alert us to illegal health products, so that we can take enforcement actions against them. We would also like to remind everyone that illegal health products can have serious health consequences. HSA will also continue to work with local and overseas partners, including INTERPOL, to expeditiously remove online listings or intercept parcel deliveries so as to protect public health and safety.”
11 Consumers are advised to be ALERT when purchasing health products:
- AVOID Dubious Products: Steer clear of products from unknown or unverified sources, especially online - they may contain harmful ingredients.
- LOOK for Reputable Sellers: Buy from trusted pharmacies or retailers with an established presence in Singapore.
- EXAMINE Claims: Be cautious and wary of products that claim to have miraculous results, or “no side effects”. In addition, consumers should always be cautious of effects that seem too good to be true, regardless of its source.
- REPORT Suspicious Products: Help protect others by reporting dangerous or dubious products to HSA.
- TRUST Medical Advice. Consult a healthcare professional if you need help managing your chronic medical conditions instead of risking your health with use of unfamiliar products.
12 HSA takes a serious stand against illegal activities involving health products. Anyone caught importing, manufacturing and/or supplying illegal health products is liable on conviction, to an imprisonment term for up to 3 years and/or fined up to $100,000.
13 Members of the public are encouraged to report any illegal activity involving illegal health products to HSA at Tel: 6866-3485 from 8.30am to 5.30pm (Monday to Friday), or e-mail: hsa_is@hsa.gov.sg.
HEALTH SCIENCES AUTHORITY
SINGAPORE
7 MAY 2026
ANNEX
Photos of illegal products and product listings detected
Online listing of unregistered contact lenses
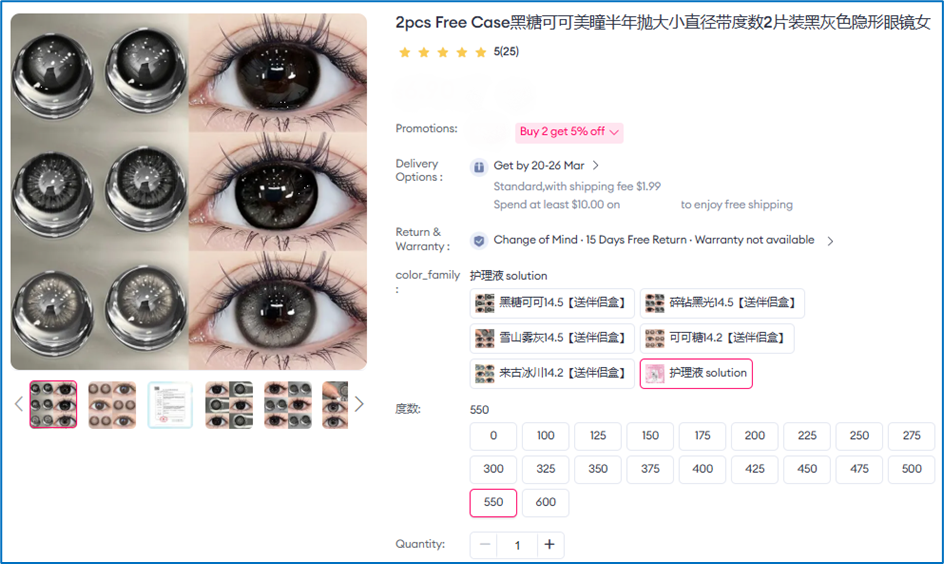
Online listing of illegal acne skin cream
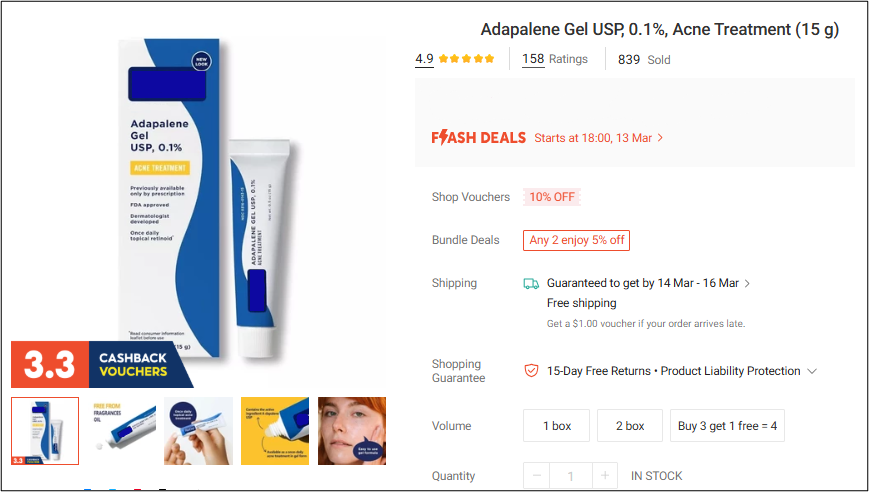
Online listing of illegal sexual enhancement medicine
Infographic

Consumer, Healthcare professional, Industry member
Published:
Subscribeto stay up to date with HSA news and regulatory updates.
Press Releases